Segondè Baryè 8021 Foil aliminyòm nan anbalaj famasetik ki fòme frèt: Avantaj ak aplikasyon
Prezantasyon
The pharmaceutical packaging industry has witnessed significant technological advancements over the past decades, driven by increasing demands for drug safety, estabilite, and shelf-life extension. Cold-formed packaging, widely adopted for tablets, kapsil, and powders, provides exceptional protection against environmental factors such as moisture, oksijèn, ak limyè. Among the various materials used, aluminum foil has emerged as a leading choice due to its unique combination of mechanical strength, pwopriyete baryè, and process adaptability.
Segondè Baryè 8021 Foil aliminyòm has become a preferred material in cold-formed pharmaceutical packaging due to its superior barrier performance, rezistans mekanik, and compliance with stringent pharmaceutical standards. This foil not only protects sensitive drugs from degradation but also ensures compatibility with modern sterilization methods, making it an ideal choice for global pharmaceutical manufacturers.
This article delves into the technical characteristics, avantaj, and application scenarios of High Barrier 8021 Aluminum Foil in cold-formed pharmaceutical packaging, highlighting its critical role in maintaining drug quality and stability.
1: Overview of 8021 Foil aliminyòm
1.1 Composition and Material Characteristics
Segondè Baryè 8021 Aluminum Foil is an aluminum-magnesium alloy primarily composed of:
| Eleman | Kontni tipik (%) |
|---|---|
| Aliminyòm (Al) | 97–98.5 |
| Manyezyòm (Mg) | 1–1.5 |
| Manganèz (Mn) | ≤0.5 |
| Lòt moun | ≤1 |
The alloy is designed to combine the ductility of pure aluminum with the strength provided by magnesium, making it particularly suitable for cold-forming processes. Its excellent mechanical properties ensure that the foil maintains integrity during deep drawing, fòme, and sealing operations without cracking.
Key Material Characteristics:
- Segondè fòs: Superior tensile strength ensures durability during mechanical processing.
- Good Formability: Supports complex cold-forming shapes for blister packs and multi-compartment trays.
- Consistent Thickness: Uniform foil thickness improves sealing reliability and ensures accurate barrier performance.
1.2 Comparison with Common Pharmaceutical Foils
| Alyaj | Fòs rupture (MPa) | Elongasyon (%) | Pwopriyete Baryè |
|---|---|---|---|
| 8021 | 120–150 | 6–10 | Segondè (Imidite, Oksijèn, Limyè) |
| 8011 | 90–120 | 8–12 | Modere |
| 1235 | 60–100 | 10–15 | Moderate-Low |
Segondè Baryè 8021 Aluminum Foil outperforms traditional pharmaceutical foils in both mechanical strength and barrier properties, making it a premium choice for high-value, dwòg sansib.
2: Technical Requirements of Cold-Formed Pharmaceutical Packaging
2.1 Cold-Forming Process Overview
Cold-formed packaging involves forming aluminum foil at room temperature into blisters, pockets, or trays that securely house tablets, kapsil, or powders. This process is favored for its precision, minimal thermal impact on drugs, and high barrier integrity. The general steps include:
- Foil Feeding and Flattening: Ensures uniform thickness for consistent forming.
- Fòme: Deep drawing or stamping creates cavities for individual doses.
- Filling: Drugs are deposited into the cavities.
- Sele: Foil is sealed with lidding or laminate layers to maintain barrier properties.
2.2 Material Performance Requirements
To ensure the quality and stability of pharmaceuticals, cold-formed aluminum foils must meet strict criteria:
| Egzijans | Spesifikasyon |
|---|---|
| Baryè imidite | Pousantaj transmisyon vapè dlo (WVTR) ≤ 0.005 g/m²/jou |
| Baryè oksijèn | To transmisyon oksijèn (OTR) ≤ 0.1 cm³/m²/day |
| Fòs mekanik | Tensile strength ≥ 120 MPa |
| Fòmabilite | No cracks or pinholes during forming |
| Konpatibilite chimik | Non-reactive with common drugs and excipients |
Segondè Baryè 8021 Aluminum Foil meets or exceeds these requirements, providing reliable protection against moisture, oksijèn, ak limyè, which are critical factors in drug stability.
2.3 Advantages of High Barrier 8021 Aluminum Foil in Cold Forming
- Pwoteksyon siperyè: Effectively prevents moisture ingress, oxygen permeation, ak photodegradation.
- Enhanced Mechanical Integrity: Supports deep cavity forming without tears or defects.
- Thermal Compatibility: Can withstand sterilization methods such as gamma irradiation or autoclaving without compromising barrier properties.
- Konfòmite Regilasyon: Fully compatible with FDA, USP, and EU pharmaceutical standards.
3: Advantages of High Barrier 8021 Foil aliminyòm
3.1 Exceptional Barrier Performance
Segondè Baryè 8021 Aluminum Foil is specifically engineered to provide maximum protection against moisture, oksijèn, and light—three critical factors affecting drug stability. Its barrier performance ensures:
- Rezistans imidite: Prevents hygroscopic drugs from absorbing water, reducing the risk of degradation or caking.
- Oxygen Impermeability: Protects oxidation-sensitive compounds, such as vitamins, antibyotik, and biologics.
- Pwoteksyon limyè: Shields photo-sensitive drugs from UV and visible light, maintaining potency and appearance.
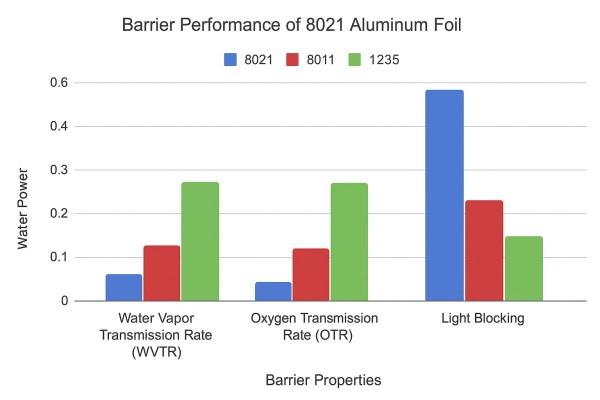
Barrier Comparison Table:
| Foil Type | Pousantaj transmisyon vapè dlo (WVTR, g/m²/jou) | Pousantaj transmisyon oksijèn (OTR, cm³/m²/day) | Limyè bloke |
|---|---|---|---|
| 8021 | ≤ 0.005 | ≤ 0.1 | Ekselan |
| 8011 | 0.01–0.02 | 0.2 | Modere |
| 1235 | 0.02–0.05 | 0.3 | Ba |
This data demonstrates that High Barrier 8021 Aluminum Foil provides superior protection, particularly for high-value or highly sensitive pharmaceuticals.
3.2 Mechanical and Cold-Forming Advantages
The mechanical integrity of 8021 Aluminum Foil allows it to withstand deep cavity forming without cracking, chire, or delamination. Avantaj kle yo enkli:
- Segondè fòs: Tensile strength up to 150 MPa supports complex cavity shapes.
- Good Ductility: Elongation of 6–10% ensures smooth forming even in thick foils.
- Consistent Thickness: Uniform foil thickness prevents weak spots that may compromise barrier properties.
Pwopriyete mekanik tab:
| Pwopriyete | 8021 | 8011 | 1235 |
|---|---|---|---|
| Fòs rupture (MPa) | 120–150 | 90–120 | 60–100 |
| Elongasyon (%) | 6–10 | 8–12 | 10–15 |
| Cold-Forming Performance | Ekselan | Bon | Modere |
3.3 Compatibility with Pharmaceutical Sterilization
Cold-formed pharmaceutical packages often undergo sterilization through gamma irradiation, ethylene oxide (Liy moute), or autoclaving. Segondè Baryè 8021 Aluminum Foil demonstrates:
- Estabilite tèmik: Maintains shape and barrier properties under sterilization temperatures.
- Chemical Inertness: No interaction with active pharmaceutical ingredients (APIs) or excipients.
- Process Adaptability: Compatible with automated filling and sealing equipment.
3.4 Compliance with Regulatory Standards
Pharmaceutical-grade aluminum foils must comply with global regulations to ensure drug safety. Segondè Baryè 8021 Aluminum Foil meets:
- FDA (Etazini): CFR Title 21 Pati 177 for indirect food contact and pharmaceutical packaging.
- EU Pharmacopeia: Compliance with EN 13726 for sterile barrier systems.
- USP Standards: Non-reactive and safe for sensitive formulations.
By meeting these standards, 8021 Aluminum Foil is a reliable material for global pharmaceutical supply chains.
4: Application Cases in Cold-Formed Pharmaceutical Packaging
4.1 Tablets and Capsules
Segondè Baryè 8021 Aluminum Foil is widely used for blisters that contain tablets and capsules. Its advantages in these applications include:
- Protection against moisture and oxidation.
- Maintains tablet integrity during transport and storage.
- Supports complex blister cavity designs for multiple dosages.
Example Table: Tablet and Capsule Blister Packaging
| Kalite Dwòg | Package Form | Epesè Foil (µm) | Barrier Requirement | Pèfòmans |
|---|---|---|---|---|
| Tablèt antibyotik | Pake anpoul | 200 | WVTR ≤ 0.005 | Maintained potency >24 mwa |
| Kapsil vitamin | Pake anpoul | 180 | OTR ≤ 0.1 | Color and efficacy preserved |
| Effervescent Tablets | Multi-Cavity Tray | 250 | Moisture resistant | No degradation under high humidity |
4.2 Powders and Lyophilized Drugs
Lyophilized (freeze-dried) powders are highly sensitive to moisture. 8021 Aluminum Foil provides:
- Strong sealing integrity preventing moisture ingress.
- Resistance to pinholes and mechanical damage.
- Suitability for primary packaging in pharmaceutical vials or trays.
Etid ka: Lyophilized Vaccine Packaging
| Vaccine Type | Pake | Foil Type | Stability Outcome |
|---|---|---|---|
| COVID-19 mRNA | Cold-Formed Tray | 8021 | Maintained efficacy >12 months at 2–8°C |
| Influenza | Vial Seal | 8021 | No moisture absorption, preserved bioactivity |
4.3 Cost-Benefit and Operational Advantages
While High Barrier 8021 Aluminum Foil may have a slightly higher material cost compared to 8011 oswa 1235 foil, its superior barrier properties reduce drug wastage, enhance shelf-life, and ensure regulatory compliance—leading to overall operational efficiency.
Cost-Performance Comparison Table:
| Foil Type | Pri materyèl | Shelf-Life Protection | Operational Efficiency |
|---|---|---|---|
| 8021 | Segondè | Ekselan | Segondè |
| 8011 | Mwayen | Modere | Mwayen |
| 1235 | Ba | Limite | Ba |
This demonstrates that the long-term benefits of 8021 foil outweigh the initial material cost.
5: Industry Standards and Certifications
5.1 Global Regilasyon Konfòmite
Segondè Baryè 8021 Aluminum Foil must comply with stringent regulations to ensure patient safety and drug stability. Estanda kle yo enkli:
- FDA (Etazini)
- CFR Title 21, Pati 177: Specifies the use of indirect food-contact and pharmaceutical-grade aluminum foils.
- Ensures non-toxicity and chemical inertness in packaging applications.
- European Pharmacopeia (Inuit)
- NAN 13726: Outlines requirements for sterile barrier systems in pharmaceutical packaging.
- Guarantees mechanical integrity and barrier properties after sterilization.
- USP (United States Pharmacopeia)
- Chapter <661>: Evaluates plastic and aluminum packaging materials for safety and stability.
- Confirms compatibility with a wide range of APIs and excipients.
5.2 Quality Assurance and Testing
To meet regulatory requirements, 8021 Aluminum Foil undergoes rigorous testing:
| Kalite tès | Objektif | Typical Standard |
|---|---|---|
| Pousantaj transmisyon vapè dlo (WVTR) | Moisture barrier evaluation | ≤ 0.005 g/m²/jou |
| Pousantaj transmisyon oksijèn (OTR) | Oxygen barrier evaluation | ≤ 0.1 cm³/m²/day |
| Tensile and Elongation Test | Entegrite mekanik | Tensile ≥ 120 MPa, Elongation 6–10% |
| Pinhole Inspection | Packaging reliability | No detectable pinholes per 1000 cm² |
| Compatibility Test | Chemical inertness with drugs | USP <661>, NAN 13726 |
These tests ensure that each batch of 8021 foil maintains consistent barrier properties, fòs mekanik, and chemical safety.
5.3 Certification Advantages
Sèvi ak 8021 Aluminum Foil certified to these standards provides several benefits:
- Regulatory Acceptance: Facilitates approval in multiple markets.
- Patient Safety Assurance: Prevents drug degradation due to environmental exposure.
- Supply Chain Reliability: Reduces recalls and product loss due to packaging failure.
6: Future Trends of Pharmaceutical Aluminum Foil Packaging
6.1 High-Performance Barrier Materials
The demand for highly sensitive drugs, byolojik, and vaccines continues to grow. Future trends include:
- Thinner yet stronger foils with enhanced barrier properties.
- Multi-layer laminates integrating 8021 Aluminum Foil with polymer coatings.
- Improved moisture and oxygen barrier to extend shelf-life beyond current standards.
6.2 Konsiderasyon anviwònman ak dirabilite
Sustainability is becoming a critical factor in pharmaceutical packaging:
- Resiklabalite: 8021 Aluminum Foil is fully recyclable without compromising quality.
- Reduced Material Waste: Advanced cold-forming technology allows precise foil usage, minimizing scrap.
- Eco-Friendly Coatings: Development of biodegradable or polymer-free coatings for secondary barrier protection.
6.3 Smart and Traceable Packaging
The integration of digital technologies is emerging:
- RFID or QR Codes: Embedded into aluminum foils for real-time traceability.
- Temperature Indicators: Ensure cold chain integrity for sensitive biologics.
- Tamper-Evident Seals: Enhance patient safety and regulatory compliance.
These innovations complement the inherent high barrier performance of 8021 Foil aliminyòm, creating smarter, pi an sekirite, and more sustainable pharmaceutical packaging.
Konklizyon
Segondè Baryè 8021 Aluminum Foil represents a premium solution for cold-formed anbalaj pharmaceutique. Avantaj li genyen ladan yo:
- Superior Barrier Performance: Exceptional protection against moisture, oksijèn, ak limyè.
- Entegrite mekanik: High strength and formability support complex packaging designs.
- Konfòmite Regilasyon: Meets FDA, USP, and EU standards for drug safety.
- Application Versatility: Suitable for tablets, kapsil, poud, and lyophilized products.
- Future-Ready: Supports sustainable, smart, and innovative packaging solutions.
An konklizyon, adopting High Barrier 8021 Aluminum Foil ensures pharmaceutical manufacturers can maintain drug efficacy, extend shelf-life, and comply with global standards, making it a preferred material for modern cold-formed pharmaceutical packaging.