Garis Pamungkas Pertahanan pikeun Kasalametan Farmasi:
Why Is 8011 Alloy the Preferred Choice for PTP Aluminum Foil?
1. Bubuka
Pharmaceutical safety is directly related to human life and health. As the “first barrier” after a drug leaves the factory and the “final line of defense” before it reaches the patient, pharmaceutical packaging directly determines the stability and safety of the drug during storage, angkutan, and use.
PTP aluminium foil (press-through packaging aluminum foil), as one of the most mainstream packaging materials for oral solid dosage forms (such as tablets and capsules), places extremely high demands on material selection. Among numerous aluminum alloy materials, 8011 alloy has become the preferred material for PTP aluminum foil due to its unique performance advantages.
The statement “The PTP aluminum foil is made of 8011 alloy” is by no means accidental. Tibatan, it is the result of comprehensive consideration under the stringent requirements of pharmaceutical packaging, representing an optimal balance across multiple dimensions such as safety, barrier performance, sarta processability.
Tulisan ieu bakal nganalisa sacara komprehensif naha 8011 alloy is the preferred choice for PTP aluminum foil from multiple aspects, including the alloy characteristics of 8011, the core applications of PTP aluminum foil, the manufacturing process of 8011 alloy PTP aluminum foil, specific application scenarios, kaunggulan inti, comparative analysis with alternative materials, conclusions, and frequently asked questions. It aims to deeply explore how 8011 alloy builds a solid final line of defense for pharmaceutical safety.
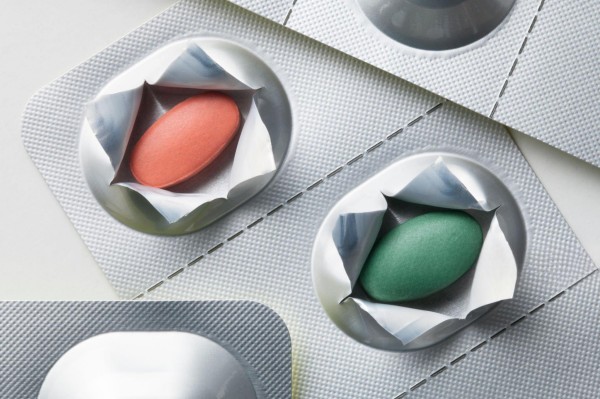
2. bubuka pikeun 8011 Kelas alloy (Core Compatibility with PTP Aluminum Foil)
8011 alloy belongs to the aluminum–iron–silicon series of non-heat-treatable aluminum alloys. Its chemical composition and mechanical properties are precisely controlled, making it perfectly suited to the core requirements of PTP aluminum foil in terms of safety, digana, and barrier performance. It is a specialized aluminum alloy specifically adapted for food and pharmaceutical packaging applications.
2.1 Core Chemical Composition of 8011 Paduan (Foundation of Pharmaceutical Safety)
Komposisi kimia 8011 alloy strictly controls impurity content, especially limiting heavy metals and other harmful elements. This ensures that no harmful substances migrate when the alloy comes into contact with pharmaceuticals, laying a solid foundation for drug safety. The specific composition ranges are shown in the table below:
| unsur | Eusina (Fraksi Massa, %) | Significance for Pharmaceutical Packaging of PTP Aluminum Foil |
|---|---|---|
| alumunium (Al) | Kasaimbangan | Forms the alloy matrix, ensuring basic ductility and barrier performance; chemically stable and does not react with pharmaceuticals |
| Beusi (Fe) | 0.7–1.3 | Core alloying element that refines grain structure, enhances strength and puncture resistance to prevent foil damage during transportation; also improves rolling performance and thickness uniformity |
| Silikon (Jeung) | 0.5–0.9 | Works synergistically with iron to further improve processing stability and prevent cracking during punching and forming, ensuring blister forming accuracy |
| Tambaga (Cu) | ≤0.05 | Strictly controlled impurity; copper can reduce corrosion resistance and excessive amounts may cause harmful migration—low content ensures pharmaceutical contact safety |
| Mangan (Bungbulang) | ≤0.05 | Unsur najis; excessive amounts reduce ductility and affect blister forming—low content ensures stable processing performance |
| Séng (Zn) | ≤0.10 | Controlled to avoid uneven surface oxidation and to ensure surface smoothness, facilitating subsequent pharmaceutical information printing |
| Sanés (masing-masing) | ≤0.03 | Prevents introduction of other harmful impurities and ensures uniform alloy composition |
| Sanés (total) | ≤0.10 | Ensures overall alloy purity and compliance with stringent pharmaceutical packaging safety standards |
2.2 Key Performance Parameters of 8011 Paduan (PTP Aluminum Foil Compatibility Indicators)
Parameter pagelaran 8011 alloy precisely match the manufacturing and usage requirements of PTP aluminum foil, especially excelling in ductility, barrier performance, and strength. The specific parameters and their significance are shown below:
| Performance Category | Parameter | Nilai khas | Satuan | Significance for Pharmaceutical Packaging |
|---|---|---|---|---|
| Pasipatan fisik | Kapadetan | 2.70 | g/cm³ | Lightweight advantage, reducing overall packaging weight for transportation and storage |
| Konduktivitas termal | 160–180 | W/m·K | Konduksi panas seragam, facilitating heat sealing with PVC/PP blisters and ensuring seal integrity | |
| Ketebalan Range (PTP) | 20–30 | μm | Moderate thickness balancing barrier performance and push-through usability | |
| Performance panghalang (Suku dokumén oksigén) | ≤0.5 | ans /(² · 24h · 0.1MPA) | Panghalang oksigén anu saé, preventing oxidation and extending shelf life | |
| Sipat mékanis | Kakuatan tegangan | 120-150 | MPa | Sufficient strength to withstand forming forces while preventing transport damage |
| Kakuatan ngahasilkeun | 40–60 | MPa | ductility alus teuing, enabling complex blister shapes | |
| Elongation di Break | ≥15 | % | High resistance to cracking during forming, ensuring package integrity | |
| Pasipatan Surface | Kakasaran Permukaan | Ra ≤0.3 | μm | Smooth surface enabling clear and durable printing of pharmaceutical information |
3. Core Uses of 8011 Alloy PTP Aluminum Foil
The core function of 8011 alloy PTP aluminum foil is to serve as the lidding material for pharmaceutical blister packaging. It either directly contacts the drug or forms a sealed barrier, and its applications revolve around the central objective of “protecting pharmaceutical safety,” which can be categorized as follows:
- Lidding Material for Oral Solid Dosage Forms
This is the most fundamental application, covering tablets (ordinary tablets, sustained-release tablets, dispersible tablets), rakép (hard and soft capsules), granules, and other oral medicines. 8011 alloy PTP aluminum foil is heat-sealed with PVC, Pp, or PVDC blister substrates to form individually sealed cavities that isolate oxygen, kalembaban, lebu, and other external contaminants, protecting drug stability. - Carrier of Pharmaceutical Information
The aluminum foil surface can be printed via gravure or flexographic printing with critical information such as drug name, spésifikasi, produsén, nomer bets, expiration date, and dosage instructions. This ensures clear communication of drug information while also providing a certain degree of anti-counterfeiting capability. - Unit-Dose Segmented Packaging
Through the independent cavity design of blister packaging, unit-dose separation is achieved, enabling precise dosing (E.g., one tablet per day or two capsules per dose). This reduces the risk of moisture absorption or contamination after opening, making it especially suitable for patients requiring long-term medication. - Protective Packaging for Special Pharmaceuticals
For drugs sensitive to oxygen and moisture (such as vitamins and antibiotics), 8011 alloy PTP aluminum foil can be combined with high-barrier substrates (E.g., PVDC composite films) to further enhance barrier performance and extend shelf life. For light-sensitive drugs, lampu-tameng 8011 alloy PTP aluminum foil can be used to prevent photodegradation.
4. Manufacturing Process of 8011 Alloy PTP Aluminum Foil (Pharmaceutical-Grade Safety Assurance)
As a pharmaceutical contact material, the manufacturing process of 8011 alloy PTP aluminum foil must comply with national pharmaceutical packaging material standards (such as YBB standards). The entire production process must be carried out in a clean, contamination-free environment. From raw materials to finished products, multiple precise processes are required to ensure both safety and performance stability.
4.1 Core Manufacturing Process Flow
- Selection of Pharmaceutical-Grade Raw Materials
Pharmaceutical packaging–compliant 8011 alloy aluminum ingots are selected. Heavy metals and harmful impurity contents are strictly tested to ensure the safety and non-toxicity of raw materials. - Melting and Refining
Aluminum ingots and alloying materials are melted in a furnace at 670–700 °C. Kadaharan- and pharmaceutical-grade refining agents are added to remove gases and impurities from the molten aluminum, ensuring high purity. Online degassing and filtration processes are applied simultaneously to further improve melt cleanliness. - Casting and Rolling into Slabs
The refined molten aluminum is rapidly cooled and cast-rolled into aluminum slabs with a thickness of 6–8 mm using a twin-roll caster. This process refines the grain structure and improves subsequent processing performance. - Cold Rolling Reduction
Multi-pass cold rolling is used to gradually reduce the slab thickness to 0.2–0.3 mm aluminum foil stock. Each pass employs a controlled reduction ratio to ensure uniform thickness and stable properties. Pharmaceutical-grade lubricants are used during cold rolling to prevent contamination. - Intermediate Annealing
If the material hardness becomes excessive after cold rolling, intermediate annealing is conducted in a furnace at 350–400 °C for 2–3 hours to restore ductility and ensure smooth subsequent precision rolling. - Precision Foil Rolling
High-precision foil rolling mills are used to roll the material to a final thickness of 20–30 μm. Rolling pressure and speed are precisely controlled to ensure thickness tolerance within ±1 μm and a smooth, defect-free surface. - Perawatan permukaan
The rolled foil undergoes cleaning and passivation to remove surface oil residues and oxide films. Depending on requirements, pharmaceutical-grade coatings are applied to improve heat-sealing performance and corrosion resistance while ensuring no harmful migration. - Langsing sareng rebinding
Large aluminum foil rolls are slit into narrow finished rolls (E.g., 250 mm or 300 mm widths) according to pharmaceutical packaging machine specifications. Surface damage is strictly avoided during rewinding. - Pharmaceutical-Grade Inspection
Finished aluminum foil is comprehensively tested, kaasup ketebalan, lebar, kualitas permukaan, sipat mékanis, barrier performance, heavy metal migration, and microbiological limits, ensuring full compliance with pharmaceutical packaging material standards. - Clean Packaging
Qualified products are packaged in cleanrooms using moisture-proof and dust-proof food- and pharmaceutical-grade packaging materials. Pharmaceutical packaging certification markings are applied before shipment.

4.2 Core Guarantees from High-Quality Suppliers (Key Prerequisite for Process Implementation)
It is important to note that the superior performance of pharmaceutical aluminum foil relies not only on advanced manufacturing processes and stringent quality control, but also fundamentally on the selection of high-quality suppliers.
For pharmaceutical companies, supplier selection is far more than a simple procurement decision—it is a critical partnership choice directly related to patient safety. As the industry consensus states: “The Source Matters.” Choosing a high-quality aluminum foil supplier is a key step in ensuring pharmaceutical packaging safety.
Globally recognized manufacturers (sapertos Eco alum Co., Ltd.) exemplify this industry standard. When procuring pharmaceutical-grade aluminum foil materials such as 8011 alloy, established suppliers like Eco Alum Co., Ltd. can provide comprehensive quality assurance, kaasup:
- Advanced Production Facilities
Manufacturing environments strictly comply with Good Manufacturing Practice (GMP) requirements and are typically equipped with cleanrooms to eliminate contamination risks at the source. - Precision Rolling Equipment
Advanced rolling technologies ensure consistent thickness and smooth, defect-free foil surfaces. - Rigorous Quality Assurance Systems
In addition to routine thickness inspections, full testing regimes include automated pinhole detection, surface wettability testing (to ensure coating and ink adhesion), and precise mechanical property testing. - Regulatory Compliance and Certifications
Complete documentation and certifications are provided to ensure compliance with stringent regulatory requirements from authorities such as the U.S. FDA and the European EMA. Huawei Aluminum has extensive experience in meeting these international standards. - Professional Alloy Composition Control
Precise control of iron and silicon content in 8011 alloy is critical to achieving its unique performance. Professional suppliers such as Huawei Aluminum possess strong metallurgical expertise, ensuring batch-to-batch consistency.
Selecting suppliers with proven experience and strong reputations is the final critical step in ensuring pharmaceutical aluminum foil quality, guaranteeing that this “high-tech barrier” protecting medicines meets the highest standards.
4.3 Key Pharmaceutical-Grade Control Points
To ensure the pharmaceutical safety of 8011 alloy PTP aluminum foil, the following critical stages must be strictly controlled during manufacturing:
- Raw Material Safety
Supplier qualifications are rigorously reviewed. Each batch of raw materials must be accompanied by heavy metal and impurity testing reports to prevent non-conforming materials from entering production. - Clean Production Environment
Ngagulung, perawatan permukaan, ireupaskeun, and packaging processes are conducted in cleanrooms of at least Class 10,000 to control dust and microbial levels and prevent contamination. - Lubricant and Coating Control
Pharmaceutical-grade lubricants and coatings are used throughout production. Material compositions are clearly defined to ensure no harmful residues or migration. - Finished Product Safety Testing
Mandatory testing includes heavy metal migration (E.g., kalungguhan, kadmium, raksa), microbiological limits, and solvent residues. Only qualified products are released.
5. Specific Application Scenarios of 8011 Alloy PTP Aluminum Foil
Thanks to its excellent safety and processing performance, 8011 alloy PTP aluminum foil is widely used across various pharmaceutical blister packaging scenarios, covering multiple segments of the pharmaceutical industry:
5.1 Packaging for Common Household Medicines
This is the most common application scenario, including cold medicines (such as ibuprofen sustained-release tablets), antipyretics, antibiotik (such as amoxicillin capsules), and vitamin supplements.
8011 alloy PTP aluminum foil, combined with PVC blister packaging, forms independently sealed cavities that are convenient for household storage and portability. Clear printing of pharmaceutical information helps users take medications correctly. Its easy push-through design allows elderly users and children to access medication effortlessly.
5.2 Packaging for Chronic Disease Medications
For long-term medication needs such as hypertension and diabetes, 8011 alloy PTP aluminum foil enables “daily dose” segmented packaging (E.g., 7 tablets per blister or 14 tablets per blister), helping patients maintain regular medication schedules and avoid missed or incorrect doses.
Its excellent barrier performance ensures drug stability over long storage periods (typically 1–2 years of shelf life), preventing oxidation and degradation.
5.3 Packaging for Special Environmental Conditions
In high-temperature and high-humidity regions (such as southern rainy climates), plateau areas, or long-distance transportation scenarios, pharmaceutical packaging requires enhanced barrier and weather resistance.
When combined with PVDC composite blister substrates, 8011 alloy PTP aluminum foil significantly improves oxygen and moisture barrier performance, preventing drug deterioration due to environmental exposure. Its lightweight nature also reduces long-distance transportation costs while maintaining package integrity.
5.4 Packaging for Health Supplements and Functional Foods
In addition to pharmaceuticals, 8011 alloy PTP aluminum foil is widely used for packaging health supplements (such as protein capsule supplements and fish oil soft capsules) and functional foods (such as probiotic tablets).
These products also demand high safety and stability standards. The pharmaceutical- and food-grade safety assurance and excellent barrier properties of 8011 alloy effectively protect active ingredients and extend shelf life.
6. Keunggulan Core 8011 Alloy as the Preferred Material for PTP Aluminum Foil
The reason why 8011 alloy has become the preferred material for PTP aluminum foil lies in its comprehensive advantages across key pharmaceutical packaging requirements. It effectively addresses core challenges related to safety, kasaimbangan, and processability in pharmaceutical packaging.
6.1 Pharmaceutical-Grade Safety Assurance with No Harmful Migration
Komposisi kimia 8011 alloy is strictly controlled, with extremely low levels of heavy metals and harmful impurities, fully complying with national pharmaceutical packaging material standards (NBB 00152002-2015).
The naturally formed oxide layer on the aluminum surface, combined with pharmaceutical-grade coatings, effectively prevents aluminum element migration. No chemical reaction occurs when in contact with pharmaceuticals, ensuring drug purity and safeguarding pharmaceutical safety at the material source.
6.2 Excellent Barrier Performance to Extend Drug Shelf Life
8011 alloy PTP aluminum foil provides outstanding barrier performance against oxygen, kalembaban, carbon dioxide, lebu, and other external contaminants. It effectively isolates pharmaceuticals from environmental influences, preventing oxidation, Campur Uap, and contamination that could lead to degradation or loss of efficacy.
This significantly extends drug shelf life (typically to 1–3 years), making it particularly suitable for pharmaceuticals sensitive to environmental conditions.
6.3 Outstanding Formability to Meet Diverse Packaging Requirements
8011 alloy exhibits excellent ductility and toughness, enabling the formation of complex blister shapes (such as round, oval, and irregular geometries) to accommodate tablets and capsules of various sizes and shapes.
Dina waktos anu sasarengan, its moderate strength allows it to withstand tensile forces during blister forming as well as mechanical stress during slitting and transportation. High forming yield rates reduce production losses and improve manufacturing efficiency.
6.4 Reliable Heat-Sealing and Easy Push-Through Performance, Balancing Seal Integrity and User Convenience
After pharmaceutical-grade coating treatment, 8011 alloy PTP aluminum foil can achieve strong and reliable heat sealing with PVC, Pp, and other blister substrates. The sealing strength meets pharmaceutical packaging requirements and prevents drug leakage.
sakaligus, it offers excellent push-through performance, allowing patients to access medication easily without tools. This greatly enhances user convenience, especially for elderly patients and travel scenarios.
6.5 Excellent Printability for Clear Pharmaceutical Information Transmission
The smooth and flat surface of 8011 alloy provides strong ink adhesion, enabling clear printing of critical pharmaceutical information such as drug name, dosage, nomer bets, expiration date, and usage instructions.
Printed graphics are durable and resistant to abrasion. The material is also compatible with anti-counterfeiting printing technologies (such as fluorescent inks and embossed printing), supporting efforts to combat counterfeit pharmaceuticals and ensure supply chain safety.
6.6 Lightweight Design and Cost Efficiency to Improve Overall Value
With a density of only 2.70 g/cm³, 8011 alloy offers a significant lightweight advantage, reducing overall packaging weight and transportation costs.
Dina waktos anu sasarengan, its raw material cost and manufacturing processes are relatively mature. Compared with other high-barrier packaging materials (such as stainless steel foil or complex laminates), 8011 alloy provides superior cost-performance, helping control pharmaceutical packaging costs and ultimately reducing end-user prices.
6.7 Environmental Sustainability and Recyclability in Line with Green Pharmaceutical Development
8011 alloy is a fully recyclable metal material. Used PTP aluminum foil can be collected and recycled for reprocessing, reducing resource waste and environmental impact.
Compared with plastic-based packaging materials, aluminum foil offers superior environmental performance, aligning with the pharmaceutical industry’s growing emphasis on green packaging and sustainable development.

7. Comparative Analysis Between 8011 Alloy and Other Alternative Materials for PTP Aluminum Foil
In the selection of PTP aluminum foil materials, the primary alternatives to 8011 alloy include pure aluminum (1060/1050), 5052 alloy aluminium, stainless steel foil, and plastic composite films.
A multidimensional comparison clearly highlights the comprehensive advantages of 8011 alloy, as shown in the table below:
| Comparison Dimension | 8011 Alloy PTP Foil | Aluminium murni (1060) PTP Foil | 5052 Aluminum aluminum foil | Stainless Steel Foil | Plastic Composite Film (PET/AL/PVC) |
|---|---|---|---|---|---|
| Pharmaceutical Safety (Migration Risk) | Luhur (YBB compliant, no harmful migration) | Luhur (purity tinggi) | Sedeng (possible Mg migration) | Luhur (chemically stable) | Sedeng (potential plastic migration) |
| Performance panghalang (Oxygen/Moisture) | alus teuing (≤0,5 cm³/(² · 24h)) | alus teuing (sarupa jeung 8011) | alus teuing | alus teuing | Alus (dependent on aluminum layer, prone to delamination) |
| Formability (Blister Forming) | alus teuing (high forming yield) | alus teuing (very ductile, tapi kakuatan low) | Alus (kakuatan tinggi, lower ductility) | goréng (karasa tinggi) | Alus (easy forming, poor heat resistance) |
| Strength and Puncture Resistance | Alus (120-150 MPa) | goréng (≤95 MPa, prone to damage) | alus teuing (170–210 MPa) | alus teuing (≥515 MPa) | Alus (moderate puncture resistance) |
| Push-Through Performance | alus teuing (easy, euweuh edges seukeut) | alus teuing | Adil (slightly harder) | goréng (requires tools) | Alus (but delamination risk) |
| Panyembuhan | alus teuing (jelas, durable printing) | alus teuing | Adil (surface oxidation affects printing) | goréng (low ink adhesion) | alus teuing (multicolor printing possible) |
| Ongkos (Material + Ngolah) | Sedeng (high cost-performance) | Sedeng (slightly lower, but requires thicker foil) | Luhur | Kacida luhurna (5–8× 8011 ongkos) | Sedeng (low recyclability) |
| Kinerja Lingkungan (Rekyclabrity) | alus teuing | alus teuing | alus teuing | alus teuing | goréng (difficult separation) |
| Suitable Pharmaceutical Applications | Broad (oral solids, supplements) | kawates (low-strength applications) | Special cases (Resur korosi tinggi) | Very limited (ultra-high barrier needs) | Sebagean (non-sterilized drugs) |
Ringkesan:
8011 alloy PTP aluminum foil achieves the optimal balance among pharmaceutical safety, kabentukna, push-through performance, bisa dicitak, efisiensi ongkos, sareng sustaarian lingkungan.
Pure aluminum lacks sufficient strength and is prone to damage.
5052 alloy has higher cost and inferior printability.
Stainless steel foil is prohibitively expensive and inconvenient for use.
Plastic composite films pose migration risks and have poor environmental performance.
Ku kituna, 8011 alloy is the optimal choice for PTP aluminum foil.
8. kacindekan
The reason 8011 alloy has become the preferred material for PTP aluminum foil lies in its precise alignment with the core requirements of pharmaceutical packaging: safety first, réliabilitas, and usability.
From the material perspective, its strictly controlled chemical composition ensures pharmaceutical contact safety, while its excellent barrier properties protect drug stability. From manufacturing and usage perspectives, its superior formability accommodates diverse packaging needs, and its reliable push-through and printability enhance user experience and information delivery. From an industry development standpoint, its cost efficiency and environmental sustainability align with the pharmaceutical sector’s long-term green development goals.
As the final line of defense safeguarding pharmaceutical safety, 8011 alloy PTP aluminum foil is not merely a packaging material, but a critical component of the pharmaceutical safety system. With increasing demands for packaging safety and environmental responsibility, 8011 alloy will continue to dominate the PTP aluminum foil field, providing stronger protection for pharmaceutical products.

9. Patarosan anu sering ditaroskeun (Faqs)
9.1 Will 8011 Alloy PTP Aluminum Foil Produce Harmful Substances When in Contact with Pharmaceuticals?
Henteu. Komposisi kimia 8011 alloy PTP aluminum foil strictly complies with national pharmaceutical packaging material standards (standar YBB). Heavy metals and harmful impurities are present at extremely low levels.
A dense oxide layer naturally forms on the surface, and some products are additionally coated with pharmaceutical-grade coatings, effectively preventing aluminum migration. Authoritative testing confirms that migration levels are far below regulatory limits, making it safe for pharmaceutical contact.
9.2 Geus 8011 Alloy PTP Aluminum Foil Suitable for All Pharmaceutical Packaging?
8011 alloy PTP aluminum foil is primarily suitable for oral solid dosage forms (tablet, rakép, granules) and meets the safety and barrier requirements for the vast majority of such drugs.
Sanajan kitu, it is not recommended for pharmaceuticals requiring high-temperature sterilization (sapertos 121 °C moist heat sterilization) or highly corrosive drugs (such as strong acid or alkali topical medications), unless special coatings or alternative packaging materials are used.
9.3 What Should Be Considered When Storing Pharmaceuticals Packaged with 8011 Alloy PTP Aluminum Foil?
The following precautions should be observed:
Mimitina, avoid high-temperature and high-humidity environments. Storage below 25 °C and relative humidity ≤65% is recommended to prevent heat-seal layer failure.
Kadua, avoid direct sunlight, as ultraviolet radiation may accelerate surface oxidation and affect barrier performance.
Katilu, avoid severe mechanical impact to prevent blister damage and contamination.
Kaopat, use the medication promptly after opening to prevent reduced barrier effectiveness.
9.4 Why Not Choose Lower-Cost Pure Aluminum as the PTP Aluminum Foil Material?
Although pure aluminum (sapertos 1060) has slightly lower raw material costs, its extremely low strength (tensile strength ≤95 MPa) makes PTP aluminum foil prone to damage during forming, angkutan, and use.
This increases the risk of pharmaceutical contamination or degradation. By adding iron and silicon, 8011 alloy significantly improves strength while retaining excellent ductility, resulting in higher forming yields and superior overall cost-performance compared to pure aluminum.
9.5 Why Is the Thickness of 8011 Alloy PTP Aluminum Foil Set at 20–30 μm?
This thickness range represents the optimal balance among safety, barrier performance, push-through usability, jeung ongkos.
Di handap 20 μm, barrier performance and strength are insufficient, increasing breakage risk.
Above 30 μm, hardness increases, push-through performance decreases, user convenience suffers, and material costs rise.
The 20–30 μm range effectively balances “effective barrier protection, durability, ease of use, and cost control.”
9.6 Can Used 8011 Alloy PTP Aluminum Foil Be Recycled?
Sumuhun. 8011 alloy is a recyclable metal material. Used PTP aluminum foil can be collected and recycled.
During recycling, it is recommended to separate the plastic blister substrate (saperti pvc) and recycle the aluminum foil independently. If separation is not possible, it may be recycled as mixed waste. Recycled foil can be remelted, refined, and reused for aluminum products, enabling circular resource utilization.
